The Science
Evaporative cooling is nature’s way of cooling. Water absorbs heat when it evaporates. The amount of heat required to evaporate 1 gallon of water is more than 8000 BTUs. How much cooling that can be realized from evaporating water is dependent on how much water can be evaporated in a given time. The more water evaporated, the more the cooling effect.
Physical Principals
The physical principal behind evaporative cooling is that water needs more energy to be in a gas state then it does in a liquid state. As water evaporates from a liquid to a gas, it absorbs energy (heat) from its surrounding environment. The result is that as water evaporates, the temperature of the water decrease, the temperature of the air absorbing the water decreases, and the temperature of anything the water is touching decreases. The amount of heat absorbed in the evaporative process is 1000 BTU’s per pound of water evaporated. The more water that evaporates, the more cooling will happen.
Practical Application
There are various evaporative cooling systems, the most common of which is a misting or fogging system. This is a system where water is injected directly into the air, in fine droplets that easily evaporate, and cool the surrounding air. More advanced than misting, are evaporative coolers. Evaporative coolers use fans to move air across wet pads and are often used to cool large spaces where humidity buildup isn’t a problem.
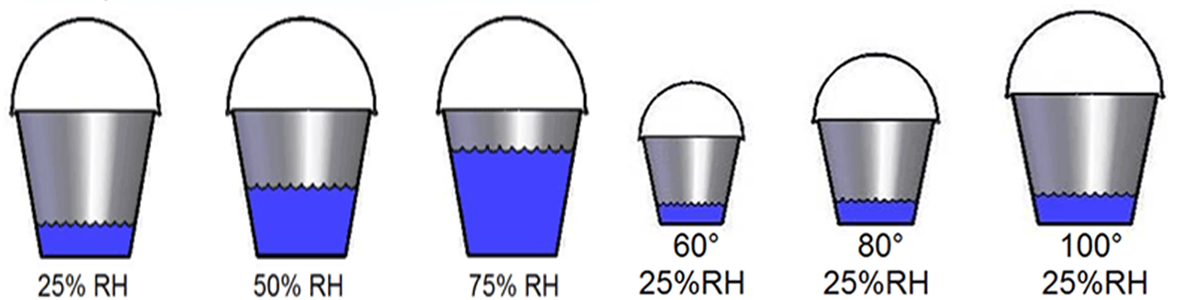
Bucket Analogy of Air's Moisture Content
If air is like a bucket, the Relative Humidity is the measurement of how full of water the bucket is.
How Much Cooling can be Realized with Evaporation?
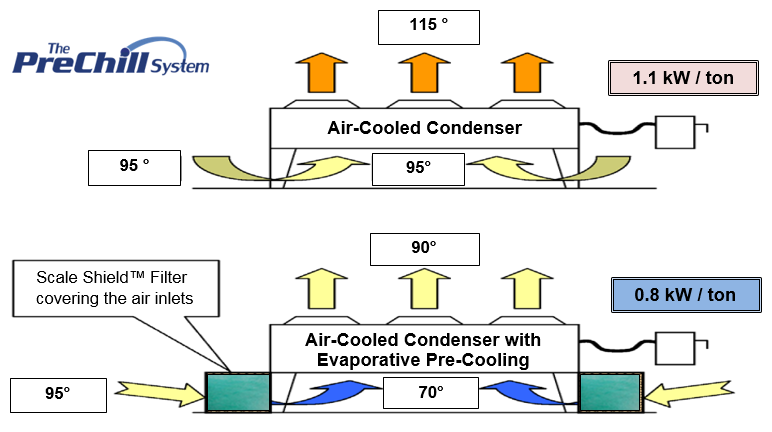
Since cooling is dependent on how much water is evaporated, the only limit to how much cooling can be done by evaporation is how much water can be held by the air. This is dependent on the temperature of the air and the Relative Humidity of the air. The Relative Humidity is a measure of how much water has already been absorbed by the air. Air at a given temperature can only go to 100% Relative Humidity.
AIR CONDITIONER EFFICIENCY
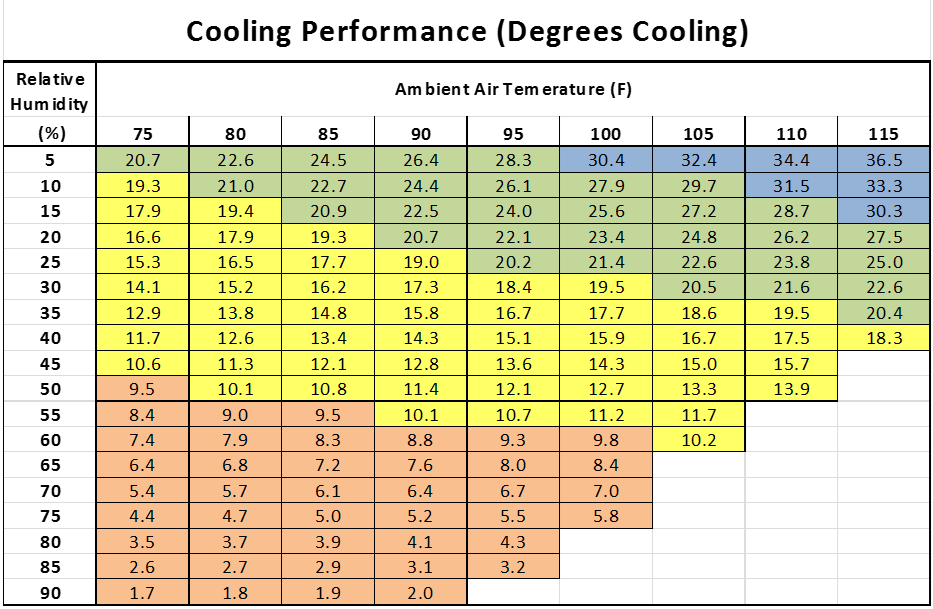
The chart shows the total amount of cooling that can be achieved through water evaporation at various humidity levels. At a relative humidity of 25%, typical in the hot dry areas of the U.S. during the Summer, the average daily cooling that evaporative cooling can provide is approx. 22 degrees F., with a maximum cooling of 25 degrees F. Lower humidity levels yield a greater cooling effect.
